Code Break
On Dec. 23, 2016, the U.S. Food and Drug Administration (FDA) approved the first successful genetic intervention for spinal muscular atrophy (SMA) — a genetic neurological condition that was then the No. 1 genetic killer of infants.
It was the first glimmer that scientists could “break the genetic code” and address underlying causes of disease, not just resign themselves to keeping babies comfortable in their short lives.
New discoveries seemed possible, but they would require a higher level of collaboration and partnership among physicians and scientists. Those at Le Bonheur, the University of Tennessee Health Science Center (UTHSC) and St. Jude Children’s Research Hospital were ready to take advantage of the opportunity.
“We know there are new discoveries in basic sciences that are close to having a real impact for children with neurologic disorders,” said Le Bonheur Neuroscience Institute Co-director James Wheless, MD. “The time is right for us to play a large role in translating these discoveries to treatments for patients as quickly as possible.”
In a new collaborative effort, Le Bonheur’s Neuroscience Institute and the St. Jude Pediatric Translational Neuroscience Initiative (PTNI) will partner to provide basic research, clinical trials and multispecialty care that will rapidly deliver new therapies to children living with genetic neurologic diseases. For many of these children, it is their first, best chance at a better quality of life — or even at surviving into adulthood.
THE VISION
A neurologist by training, J. Paul Taylor, MD, PhD, director of the St. Jude Pediatric Translational Neuroscience Initiative, had experienced firsthand the pessimistic view that neurologic diseases were untreatable. But he saw this starting to change.
“I could see that a revolution was coming with many therapies in the works for previously untreatable diseases,” he said. “So, we designed the Pediatric Translational Neuroscience Initiative to leverage and build upon the experience and infrastructure of St. Jude. Our aim was to address catastrophic pediatric neurological disorders and speed up the time from basic research to therapeutics.”
Genes for many pediatric neurological diseases were identified in the last 35 years, and the gene responsible for SMA was uncovered in 1995. Years of basic science discovery followed to determine what mutations occurred, the mechanisms of the disease and how to deliver gene-targeted therapies. But this plethora of basic research was not being translated quickly into treatments for children and adults, Taylor said.
To bridge the gap between basic research and treatments, Taylor knew he already had a partner with clinical care expertise in Memphis — Le Bonheur’s Neuroscience Institute and its renowned epilepsy program.

J. Paul Taylor, MD, PhD, is director of St. Jude Children’s Research Hospital’s Pediatric Translational Neuroscience Initiative and has worked with Le Bonheur’s Neuroscience Institute to create a new collaborative framework to bring treatments to children with neurologic diseases.
In the past few years, Le Bonheur’s Chief Neurologist Wheless, Taylor and Le Bonheur Pediatrician-in-Chief Jon McCullers, MD, who also serves as senior executive associate dean of Clinical Affairs at UTHSC, explored a new collaborative framework. By working together, they hope to leverage the strengths of each institution — translating basic research into treatments or even cures for children with devastating genetic neurologic diseases as quickly as possible.
“The St. Jude oncology model is unparalleled in its ability to conduct clinical trials for rare diseases that bring the latest treatments to children around the globe,” said Wheless. “And we have the clinical care expertise and programs to support children with neurological disease and conduct clinical trial protocols specific to those diseases.”
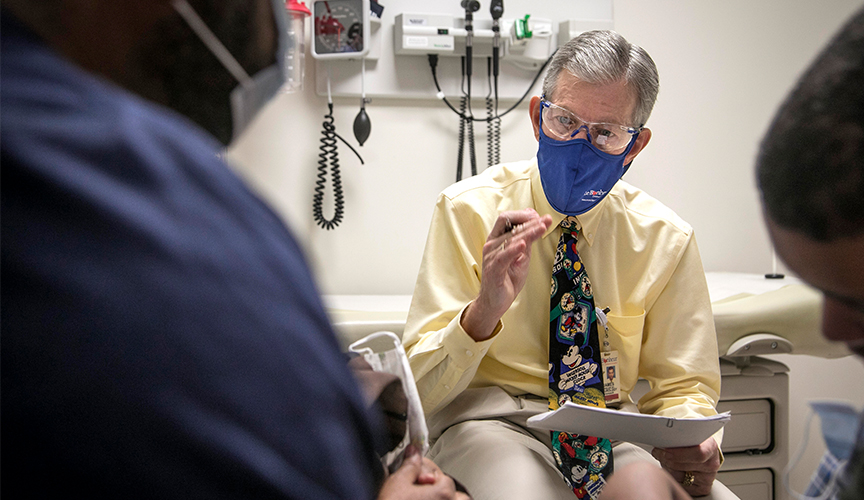
Le Bonheur’s Neuroscience Institute provides the clinical care expertise and programs to support children with neurological disease as a part of the research partnership with St. Jude. Above, Co-director of the Neuroscience Institute James Wheless, MD, sees a patient in neurology clinic.
The collaborative effort will initially focus on two major categories of neurological disease — neuromuscular conditions, including SMA and Duchenne muscular dystrophy (DMD), and genetic epilepsies such as Dravet syndrome. The existing infrastructure for clinical trials through the Children’s Foundation Research Institute at Le Bonheur allows a unique focus on rare diseases with smaller patient populations.
“Our goal is fluid integration and doing the best by the patient,” said Wheless. “We will focus on translating research into cures as quickly as possible through clinical trials and high-level clinical care.”
THE RESEARCH
The collaborative research effort will focus on moving discoveries across the continuum from basic science to clinical trial protocols conducted at both St. Jude and Le Bonheur. Through PTNI, basic research will be led by Peter McKinnon, PhD, in the newly-formed St. Jude Center for Pediatric Neurological Disease Research. Clinical research will be led by Pediatric Neurologist Richard Finkel, MD, through the St. Jude Center for Experimental Neurotherapeutics. Finkel was recruited to St. Jude in 2020 to lead the clinical arm of the center. Finkel is an international expert in SMA and has more than 30 years in translational research into the biology and treatment of neurological disorders.
The goal: accelerate the translation of that research into therapies for kids.
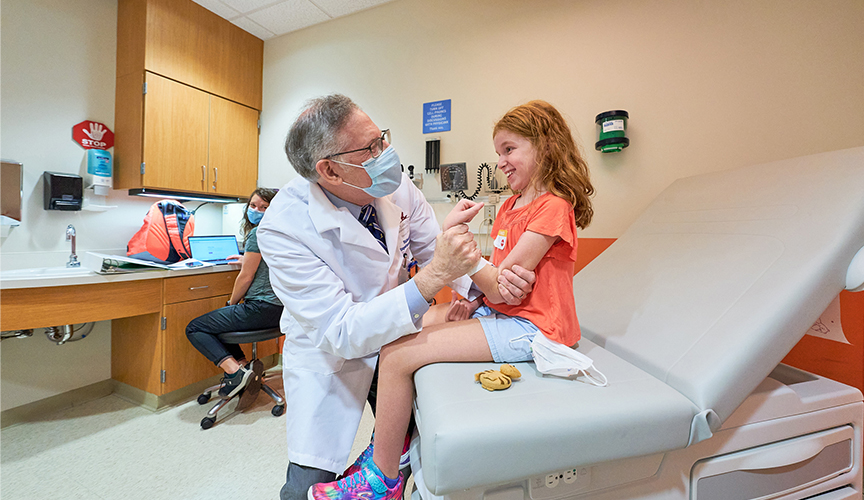
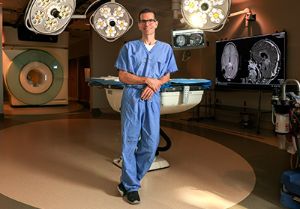
Pediatric Neurologist Richard S. Finkel, MD, (pictured above) director of St. Jude’s Center for Experimental Neurotherapeutics, will conduct clinical trial protocols at Le Bonheur’s MDA/Neuromuscular Clinic. Finkel is recognized as a pioneer of many of the clinical trials leading to FDA approval of the first genetic therapy for SMA.
The collaboration will also have a unique opportunity to develop, participate in and coordinate the earliest clinical trials for genetic therapies at Le Bonheur and St. Jude. The collaborators will work with external stakeholders, including pharma and biotech companies, to provide insights on the best ways these trials should be conducted.
“Important work is behind all clinical trials. That’s not as flashy as being the first therapy for a disease,” said Taylor. “Some of the trials will work to optimize how therapies are used or provide readiness for clinical trials by identifying biomarkers, registries and severity scales that are necessary for a trial to even take place.”
Wheless has already initiated protocols for clinical trials at Le Bonheur for genetic epilepsies, through its well-established epilepsy program. One of the most imminent treatments that will be a focus of the collaboration is a gene-targeted treatment for Dravet syndrome.
“We’re already participating in trials for novel genetic treatments for Dravet to determine if we can give patients an infusion to tweak and repair their genetic code,” said Wheless.
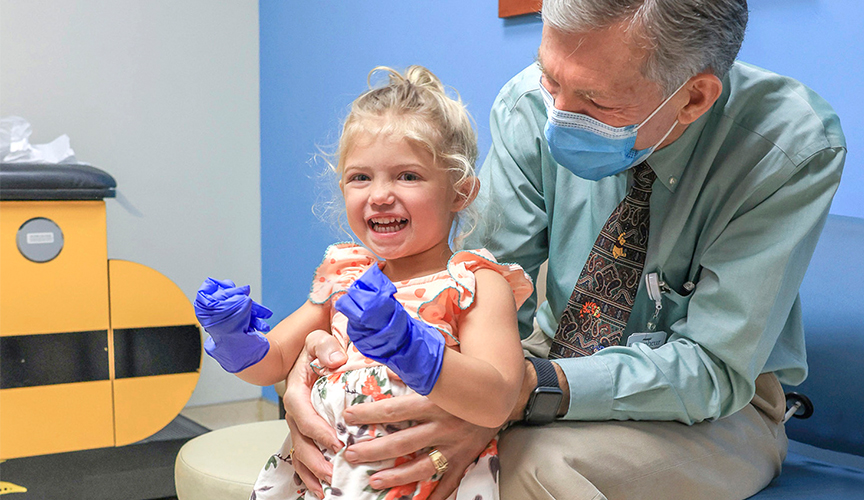
Le Bonheur Neuroscience Co-director James Wheless, MD, sees 3-year-old McCall Wright in neurology clinic. McCall is one of many children with Dravet syndrome who relies on clinical trials held at Le Bonheur and is waiting for a gene-targeted treatment for the syndrome to become widely available.
Neuromuscular disease clinical trials will be conducted out of Le Bonheur’s Muscular Dystrophy Association (MDA)/Neuromuscular Clinic. Led by Le Bonheur Neurologist Elena Caron, MD, the clinic will host clinical trials at Le Bonheur and provide expert clinical care in coordination with Finkel, who is recognized as a pioneer of many of the clinical trials leading to FDA approval of the first genetic therapy for SMA.
“Our ultimate goal is to combine what works best to push the envelope on moving treatments forward,” said Wheless. “Some protocols will take place at St. Jude, and some will integrate at Le Bonheur. But no matter what, we’re all talking and collaborating to do what’s best for this patient population.”
A Beacon of Hope: Le Bonheur participates in clinical trial for Dravet syndrome
- Dravet syndrome, a rare genetic epilepsy previously only managed by treating symptoms, causes seizures and developmental delays. Thanks to a new clinical trial, Le Bonheur is investigating a novel potential therapy to address the genetic root of Dravet syndrome. This is the first disease-modifying therapy to ever be in clinical trials for Dravet syndrome.
- A defective SCN1A gene, which controls production of a specific protein needed for movement of sodium within brain cells that is critical to proper brain function, causes Dravet syndrome. Where most people have two functioning copies of this gene, children with Dravet have only one. The new, genetically-based therapy works by modifying the production of the functioning gene to produce up to twice as much protein in an effort to restore protein levels to a more functional degree.
- The delivery system is called TANGO — targeted augmentation of nuclear gene output — and is delivered into the spinal fluid via lumbar puncture.
- “Potential treatments like this are a paradigm shift — in the past we could only treat the symptoms,” said Wheless. “Now we can target the underlying genetic issue.”
THE CARE
Patients who come to Memphis to participate in clinical trials on either campus have a range of unique clinical care needs.
“Patients undergoing protocols for new therapies have clinical needs that Le Bonheur can partner with St. Jude to handle,” said Wheless. “We have the multispecialty clinics already well established that can handle the plethora of needs these patients have.”
For example, Finkel oversees St. Jude protocols at Le Bonheur’s MDA/Neuromuscular Clinic for children who have clinical needs beyond the scope of the trial. Since this clinic is already set up with Le Bonheur providers from multiple specialties, patients can participate in a trial and have medical needs related to their condition coordinated in one visit.
Patients will also be screened for eligibility in the Le Bonheur clinics for any of the protocols underway in the collaboration. Whether the protocols take place through Le Bonheur or St. Jude, the children will be cared for according to what best meets their needs. In addition to clinical care, Le Bonheur has the wide array of neuroimaging techniques needed to provide the best imaging possible for these conditions.
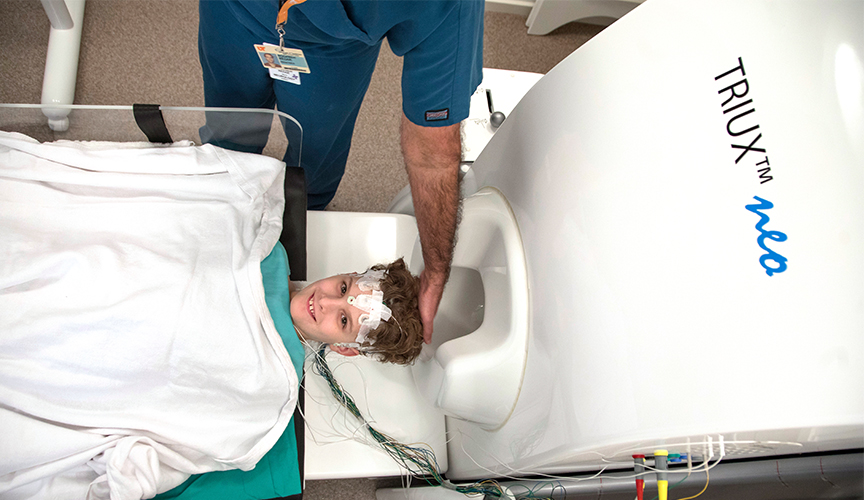
Le Bonheur will handle clinical care for children who are a part of clinical trials through the research partnership. In addition, Le Bonheur provides the neuroimaging needed for their care, including the latest generation of magnetoencephalography (MEG) technology – TRIUX™ Neo.
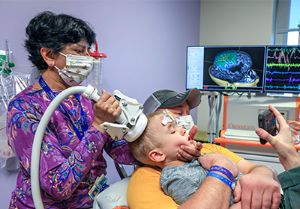
“We are pleased to be able to share our high-level technology resources,” said Wheless. “With transcranial magnetic stimulation (TMS), high-density EEG (hdEEG) and the latest generation of magnetoencephalography (MEG), we can provide cutting-edge brain imaging and analysis of brain connections for all children who are treated through this collaboration.”
THE FUTURE
Taylor and Wheless say they hope the collaboration ultimately moves the needle for pediatric neurology therapies in Memphis, the country and the world. The partnership will bring more complex patients to Le Bonheur’s campus, elevating research and clinical discoveries to improve the lives of children with devastating neurological disorders.
Furthermore, this collaborative effort also has implications for patients down the road as they begin to outlive their current life expectancies.
“With many of these diseases, children die young or have significant development issues,” said McCullers. “If we’re able to significantly treat these diseases, we have an entirely new set of issues to address if they live to adulthood that we haven’t seen before.”
The hope is that these forthcoming therapies will change thinking in the medical community that neurological diseases are untreatable.
“The collaboration has the potential to enhance care and research protocols on both sides of the street,” said Wheless. “We’re fortunate to partner with an organization where we can raise standards of care together and challenge each other to discover, test and deliver the most innovative care for the patients who depend on us.”
Genetic Epilepsy: Finding a Cure
In a recent episode of Le Bonheur's Peds Pod, Co-director of the Neuroscience Institute James Wheless, MD, explains the difference between genetic epilepsies and other epilepsies children might have, how parents can find out if their child has a genetic epilepsy and treatment options available. Listen at lebonheur.org/podcast or wherever you get your podcasts.

Help us provide the best care for kids.
Le Bonheur Children's Hospital depends on the generosity of friends like you to help us serve 250,000 children each year, regardless of their family’s ability to pay. Every gift helps us improve the lives of children.
Donate Now