Pulmonology
New Therapy for Peanut Allergy

The estimated prevalence of peanut allergy in the United States is 0.6% with between 1.5-3% prevalence estimates in the western world. The prevalence appears to be increasing, and reactions can be severe and result in death. Moreover, allergic reactions to peanuts cause the majority of food allergy-related deaths. Treatment options for peanut allergy are limited, and the main strategy is avoidance. However, peanuts are very common in food products, and trace amounts can be found in foods due to cross contamination making it difficult to avoid.
Allergist and Immunologist Jay Lieberman, MD, is leading efforts at Le Bonheur to treat peanut allergy by participating in studies such as PALISADE (Peanut Allergy Oral Immunotherapy Study for Desensitization in Children and Adults) and follow-up studies. These studies are designed to test a peanut-derived immunotherapy agent that desensitizes subjects to peanut allergen. The targeted outcome is to prevent severe reactions with small exposures. To date, the trials have shown a decrease in overall reactions to exposures, as well as decreased severity of reactions.
Clinical Trials for Cystic Fibrosis
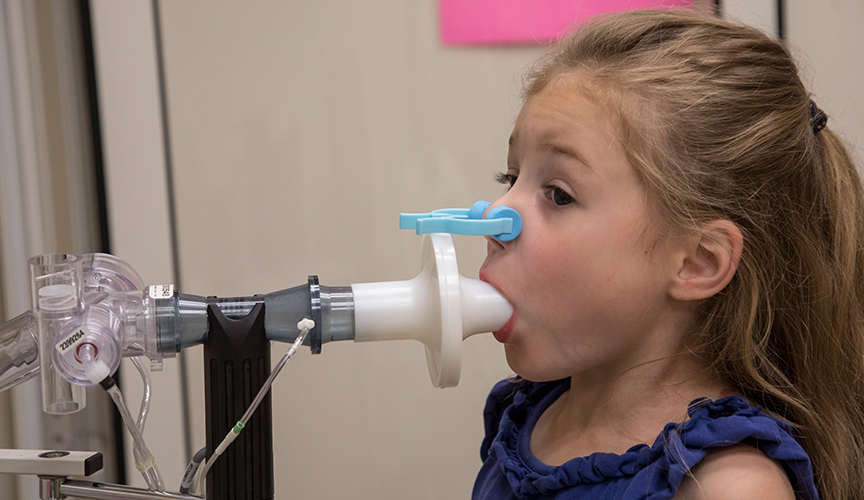
Pulmonologists Patricia Dubin, MD, and Tonia Gardner, MD, are conducting cystic fibrosis-related modulator research with participation in the state-of-the-art modulator studies. Cystic fibrosis is an inherited, multi-organ disease, and the current life expectancy is only 37 years old.
The fundamental disease-causing problem is dysfunction in an ion channel (CFTR) caused by a genetic mutation. CFTR mutations can range from very severe with essentially no CFTR being produced to mild with reasonable amounts of dysfunctional CFTR produced. These functional differences are represented by hundreds of different mutations. As a result, CFTR at the genetic level cannot yet be corrected.
Current studies have focused on developing drugs that increase CFTR levels and others that improve the function. Combinations of these modulator drugs are needed to improve CFTR function. Presently, Dubin and Gardner are leading clinical trials at Le Bonheur of new compounds to improve CFTR function and minimize undesirable side effects.
Help us provide the best care for kids.
Le Bonheur Children's Hospital depends on the generosity of friends like you to help us serve 250,000 children each year, regardless of their family’s ability to pay. Every gift helps us improve the lives of children.
Donate Now